∆Gf = ∆Hf - T.∆Sf
∆Hf and ∆Sf can be approximated by various formulae with parameters fitted to calorimetry data.
From ∆G, derive the equilibrium constant via the identity:
∆G = -RT ln KeqClick on the reactants or products in the chemical equation below to choose different species. You need to balance the equation yourself; the garbage-in, garbage-out principle applies.
Estimates are valid only for temperatures within the intersection of ranges of all involved molecules' thermochemistry model formulae. For most species supported here that range is 298K - 6000K.
Take note of the states of matter of the available substances. They may not always correspond with STP. The brotherhood of the benzene ring will know what you did.
Ammonia
 NH3NH3 (g)
NH3NH3 (g)
 NH3NH3 (g)
NH3NH3 (g)
Steam
 H2OH2O (g)
H2OH2O (g)
 H2OH2O (g)
H2OH2O (g)
Water
 H2O(l)H2O (l)
H2O(l)H2O (l)
 H2O(l)H2O (l)
H2O(l)H2O (l)
Carbon dioxide
 CO2CO2 (g)
CO2CO2 (g)
 CO2CO2 (g)
CO2CO2 (g)
Carbon monoxide
 COCO (g)
COCO (g)
 COCO (g)
COCO (g)
Carbon (graphite)
 C(graphite)C (s, graphite)
C(graphite)C (s, graphite)
 C(graphite)C (s, graphite)
C(graphite)C (s, graphite)
Methanol
 CH3OHCH3OH (g)
CH3OHCH3OH (g)
 CH3OHCH3OH (g)
CH3OHCH3OH (g)
Formaldehyde
 CH2OCH2O (g)
CH2OCH2O (g)
 CH2OCH2O (g)
CH2OCH2O (g)
Sulfur dioxide
 SO2SO2 (g)
SO2SO2 (g)
 SO2SO2 (g)
SO2SO2 (g)
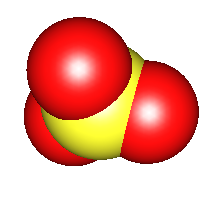
Sulfur trioxide
 SO3SO3 (g)
SO3SO3 (g)
 SO3SO3 (g)
SO3SO3 (g)
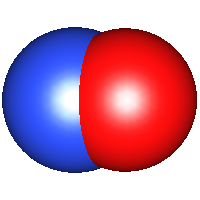
Nitric oxide
 NONO (g)
NONO (g)
 NONO (g)
NONO (g)
Nitrogen dioxide
 NO2NO2 (g)
NO2NO2 (g)
 NO2NO2 (g)
NO2NO2 (g)
Hydrogen peroxide
 H2O2H2O2 (g)
H2O2H2O2 (g)
 H2O2H2O2 (g)
H2O2H2O2 (g)
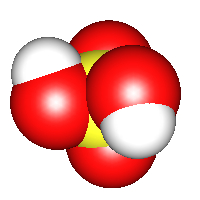
Sulfuric acid
 H2SO4H2SO4 (g)
H2SO4H2SO4 (g)
 H2SO4H2SO4 (g)
H2SO4H2SO4 (g)
Nitric acid
 HNO3HNO3 (g)
HNO3HNO3 (g)
 HNO3HNO3 (g)
HNO3HNO3 (g)
Hydrochloric acid
 HClHCl (g)
HClHCl (g)
 HClHCl (g)
HClHCl (g)
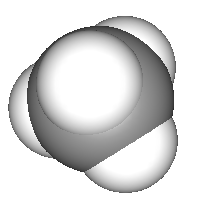
Methane
 CH4CH4 (g)
CH4CH4 (g)
 CH4CH4 (g)
CH4CH4 (g)
Ethene
 C2H4C2H4 (g)
C2H4C2H4 (g)
 C2H4C2H4 (g)
C2H4C2H4 (g)
Propane
 C3H8C3H8 (g)
C3H8C3H8 (g)
 C3H8C3H8 (g)
C3H8C3H8 (g)
Propene
 C3H6C3H6 (g)
C3H6C3H6 (g)
 C3H6C3H6 (g)
C3H6C3H6 (g)
Nitrogen
 N2N2 (g)
N2N2 (g)
 N2N2 (g)
N2N2 (g)
Hydrogen
 H2H2 (g)
H2H2 (g)
 H2H2 (g)
H2H2 (g)
Oxygen
 O2O2 (g)
O2O2 (g)
 O2O2 (g)
O2O2 (g)
Chlorine
 Cl2Cl2 (g)
Cl2Cl2 (g)
 Cl2Cl2 (g)
Cl2Cl2 (g)
Calcium oxide
 CaOCaO (s)
CaOCaO (s)
 CaOCaO (s)
CaOCaO (s)
Calcium carbonate (calcite)
 CaCO3CaCO3 (s)
CaCO3CaCO3 (s)
 CaCO3CaCO3 (s)
CaCO3CaCO3 (s)
Iron (liquid)
 Fe(l)Fe (l)
Fe(l)Fe (l)
 Fe(l)Fe (l)
Fe(l)Fe (l)
Iron (solid)
 Fe(s)Fe (s)
Fe(s)Fe (s)
 Fe(s)Fe (s)
Fe(s)Fe (s)
Hematite
 Fe2O3Fe2O3
Fe2O3Fe2O3
 Fe2O3Fe2O3
Fe2O3Fe2O3
Magnetite
 Fe3O4Fe3O4
Fe3O4Fe3O4
 Fe3O4Fe3O4
Fe3O4Fe3O4
Copper (liquid)
 Cu(l)Cu (l)
Cu(l)Cu (l)
 Cu(l)Cu (l)
Cu(l)Cu (l)
Copper (solid)
 Cu(s)Cu (s)
Cu(s)Cu (s)
 Cu(s)Cu (s)
Cu(s)Cu (s)
Cupric oxide
 CuO(s)CuO (s)
CuO(s)CuO (s)
 CuO(s)CuO (s)
CuO(s)CuO (s)
Some ads would go here if you were connected to the Internet.
Or if you weren't using an ad blocker. Or something.
It's just a placeholder, dammit.